Table of Contents
Myosin Definition
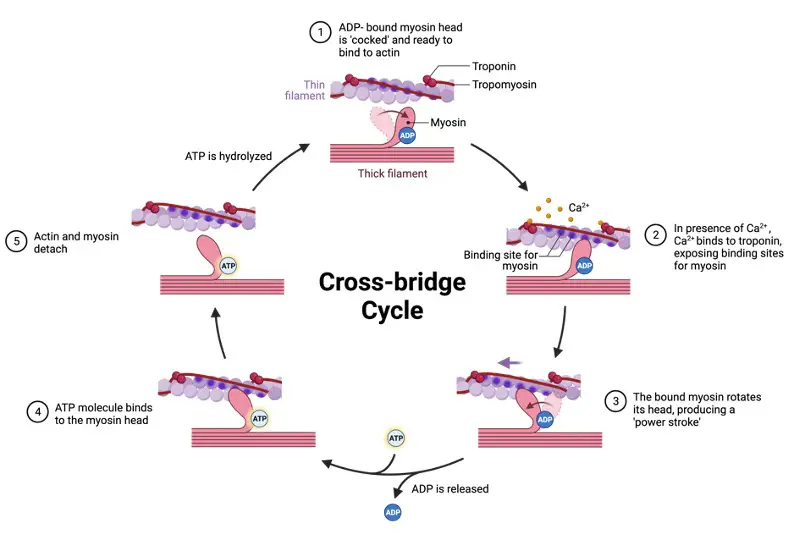
Myosin is a group of motor ATPases that interact with actin filaments are called f actin filaments. Myosin is derived from the Greek words muós, mûs (“muscle”) plus—in (“relating to muscle”).
What is Myosin?
Muscle contraction is made possible by myosin, a family of motor proteins. They may also be found as motor proteins in other cells, including amoebae and macrophages, where they are engaged in several motility processes.

The ability to bind to actin and the activity of the ATPase enzyme are two of their most important features. The head, neck, and tail domains make up the vast majority of them. The portion of myosin that binds to actin is called the head. Myosin’s light chains attach to the neck domain, which acts as a binding site. Other molecules and/or other myosin subunits interact with the tail domain.
Myosin II is a kind of myosin present in muscle cells that is involved in muscular contraction.
The number of various myosins being reported is growing. Myosin I is a motor protein that is found all over the body. It’s a globular monomeric molecule with a low molecular weight (111-128 kD) that may connect with membranes and transport membrane vesicles through microfilaments. It was identified in the cytoplasm of protozoa, acanthamoeba, and dictyostelium.
Brush boundary myosin I is a single-headed myosin that connects the membrane to the microfilament core in the microvilli of vertebrate intestinal epithelial cells. A single 119kD heavy chain and numerous (3 or 4) calmodulin light chains are present. The C terminal domain of the heavy chain bonds to acidic phospholipids.
Myosin II is a kind of myosin present in muscle cells that is involved in muscular contraction. The sarcomeric myosin that self-assembles into bipolar thick filaments is the traditional sarcomeric myosin. Myosin II is a multimeric protein (440 kD) that has two heavy chains (200 kD) and two pairs of light chains in each hexamer (17-22 kD).
Myosin II has a wide range of characteristics that differ between species and tissues. Cytoplasmic myosin II is a hexameric family of myosin-like proteins that interact with microfilaments to create force. The self-assembled filaments are shorter than those of the sarcomere, with two heavy chains (up to 240 kD) and two pairs of light chains (15-20 kD).
The result of the MYO2 gene is an unusual myosin from yeast that is involved in polarised secretion. MYO2 is possibly related to mouse dilute myosin and the vertebrate brain p190 protein. Scallop myosin is more like sarcomeric myosin than non-sarcomeric myosins because it is directly calcium controlled (via regulatory and necessary light chains).
Smooth muscle myosin is made up of two 200-kD heavy chains, two regulatory 20-kD light chains that can be phosphorylated, producing a conformational change that activates the myosin, and two 17-kD light chains. Myosin III, Myosin IV, Myosin V, Myosin VI, Myosin VII, Myosin VIII, Myosin IX, Myosin X, and others are some of the other myosins.
Myosin Citations
- The Structure of Acto-Myosin. Adv Exp Med Biol . 2020;1239:41-59.
- Myosin Structures. Adv Exp Med Biol . 2020;1239:7-19.
- Figures are created with BioRender.com







