Pyrimidine Definition
Pyrimidines are made up of carbon and nitrogen atoms and form a six-membered ring, thus they are known as aromatic compounds. All the pyrimidine derivatives including nitrogenous bases also referred to as pyrimidines.
Nitrogenous bases are of two types, purines and pyrimidines that collectively form the building blocks of both DNA (deoxyribonucleic acid) and RNA (ribonucleic acid). The organic compound pyrimidine forms pyrimidine bases through the addition of various functional groups.
There are three pyrimidines found in DNA and RNA, the thymine found in only DNA and the uracil only found in RNA whereas cytosine occurs in both DNA and RNA.
Pyrimidine Structure
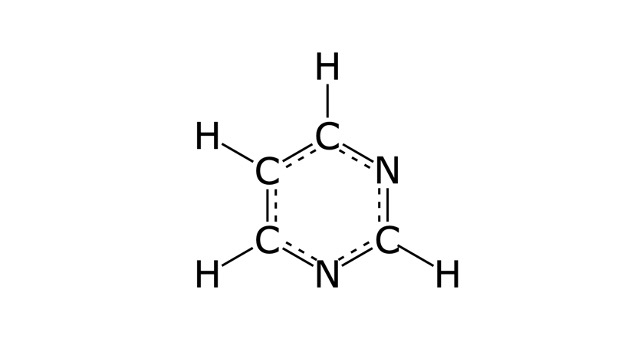
The structure of pyrimidine is made up of two nitrogen atoms and four carbon atoms. Each carbon has a hydrogen atom attached to it. Alternating double and single bonds connect the carbon and nitrogen atoms that allow resonance, or aromaticity to the molecule. The aromatic nature of pyrimidines makes them stable molecules.
The molecule derives its structure by adding functional groups. The derivatives also consist of a six-membered ring, but the addition of few atoms in nucleic acids can modify the structure to complex forms such as in drugs and vitamins.
Structure of Nitrogenous Bases
The aromatic compound pyrimidine modifies into three pyrimidine bases, thymine (T), cytosine(C), and uracil (U). The structure of these bases consists of two nitrogen atoms and four carbon atoms along with the six-membered ring, but instead of being an aromatic ring with alternating single and double bonds, they possess a carboxyl (ketone) group on the 2’carbon tom.
Therefore, a bond removes from the structure and a total of two double bonds and four single bonds are present in the structure. The other three nitrogenous bases also have various functional groups at their 4’ carbon.
Thymine and uracil have a ketone group attached, whereas cytosine has an amino group. A methyl group is also attached to the 5’carbon of thymine. In T and U, the addition of another ketone results in the removal of another double bond from the ring, and only one double bond remains in the ring.
However in cytosine, two double bonds are present but there are only two bonds to the 1’ nitrogen in all three pyrimidine bases, where the nitrogenous base attaches to the sugar and form nucleoside. The DNA and RNA also consist of several modified pyrimidines.
Several processes such as oxidation, methylation, amination, or the addition of other functional groups such as aldehydes, thioketones, and alcohols can alter the nucleotides. The modifications also cause deleterious effects in the organisms such as altering gene expression. In snRNA, the modifications are more prevalent.
Pyrimidine Function
All the pyrimidine derivatives including pyrimidine are ubiquitous. All the nucleic acids, vitamins, amino acids, antibiotics, alkaloids, and a variety of toxins have pyrimidines in their structure. Thus they play various important roles and functions such as help in the production of amino acids and proteins, contributes to an organism’s health, provide vital nutrients, boost the immune system and destroy the cell.
For example, neurotoxin tetrodotoxin is found in several species including the Japanese pufferfish, the blue-ringed octopus, and the orange-bellied newt is a pyrimidine derivative. The drug is used to prevent the transmission of nerve signals that may cause paralysis and death.
Pyrimidine derivatives also have a significant role in drug development. A wide variety of pharmaceuticals including general anesthetics, anti-epilepsy medicines, anti-malaria medicines, drugs to treat high blood pressure and HIV medications consists of pyrimidines derivatives.
Function of Nitrogenous Bases
The genetic material, i.e DNA or RNA composed of three pyrimidine nitrogenous bases, and two purine bases. They have two important functions: to pass information within the generations through replication, mitosis, meiosis, and also pass the information between different organisms through horizontal gene transfer, and to encode genes.
The DNA passes into daughter cells before passing into the offspring. The information passes in a semi-conservative manner and takes advantage of the strict rules for the nitrogenous base pairing, which can be described by Chargaff’s rules.
According to the rules, the single-ringed molecules (pyrimidines) binds or make pairs with double-ringed purines due to which the DNA maintains a constant width along the length of the molecule. In nucleic acid, the specific purines can bind to specific pyrimidines, such as the cytosine only pair with guanine, and thymine and uracil both will bind with adenine.
There are three hydrogen bonds between cytosine and guanine and two hydrogen bonds between adenine and thymine respectively. A parent molecule of a DNA strand act as a template during DNA replication.
The strand is copied by several enzymes and form an anti-parallel strand according to Chargaff’s rules. The strands of DNA and RNA are formed by the nitrogenous bases and the nucleotides that are composed of coding and non-coding regions.
Proteins are formed by the translation of the coding regions by the process called translation. The process starts from transcription or the formation of an RNA intermediary, which is later followed by translation, in which the mRNA script forms polypeptide chains.
The non-coding region cannot be translated into proteins, but they perform many other important functions, such as regulation, and encode molecules such as rRNA and tRNA, which further participate in transcription and translation.
Pyrimidine Citations
- Fused and Substituted Pyrimidine Derivatives as Profound Anti-Cancer Agents. Anticancer Agents Med Chem . 2021;21(7):861-893.
- A non-proliferative role of pyrimidine metabolism in cancer. Mol Metab . 2020 May;35:100962.
- Recent Development of Pyrimidine-Containing Antimicrobial Agents. ChemMedChem . 2020 Oct 19;15(20):1875-1886.